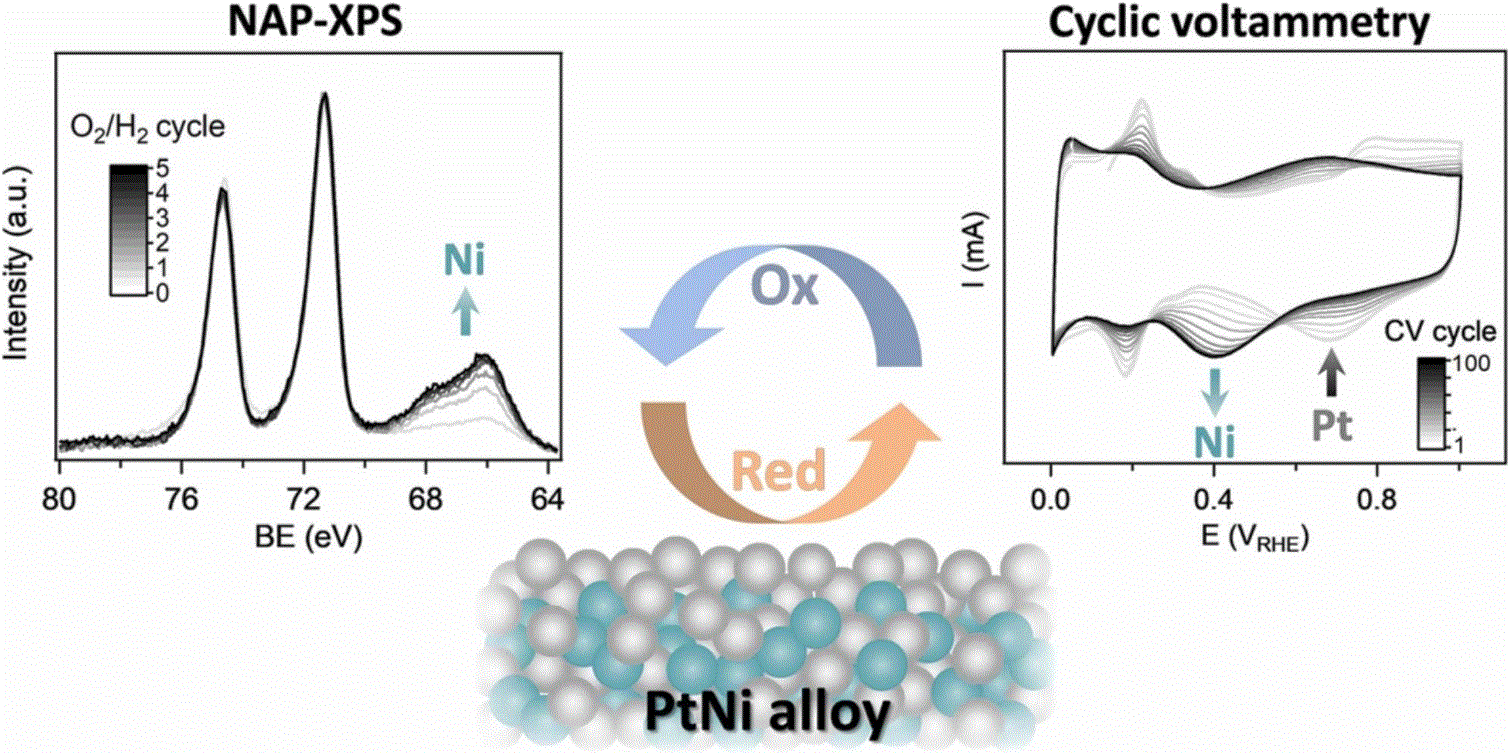
Platinum-based bimetallic alloys possess unique activities exceeding those of pure platinum. Nevertheless, as complex multi-component systems, they suffer from surface structural reorganization under operating conditions, strongly affecting their lifetime performance. This work reports an in situ electrochemical and spectroscopic study of surface compositional changes in a PtNi catalyst during repetitive oxidation/reduction cycles, simulating inherent working conditions for numerous redox reactions. Using cyclic voltammetry and near-ambient pressure X-ray photoelectron spectroscopy, quantitative surface characterization under realistic electrified liquid and gaseous environments are obtained and correlated. We show that regardless of the operating environment, PtNi undergoes a significant and irreversible change in compositional profile reflected in surface nickel enrichment and consequent catalyst deactivation, exemplary confirmed using a methanol electrooxidation reaction.
More information can be found here: https://www.sciencedirect.com/science/article/abs/pii/S0926337322012693.
© 2021 Matematicko-fyzikální fakulta Univerzity Karlovy.
Všechna práva vyhrazena. | Cookies