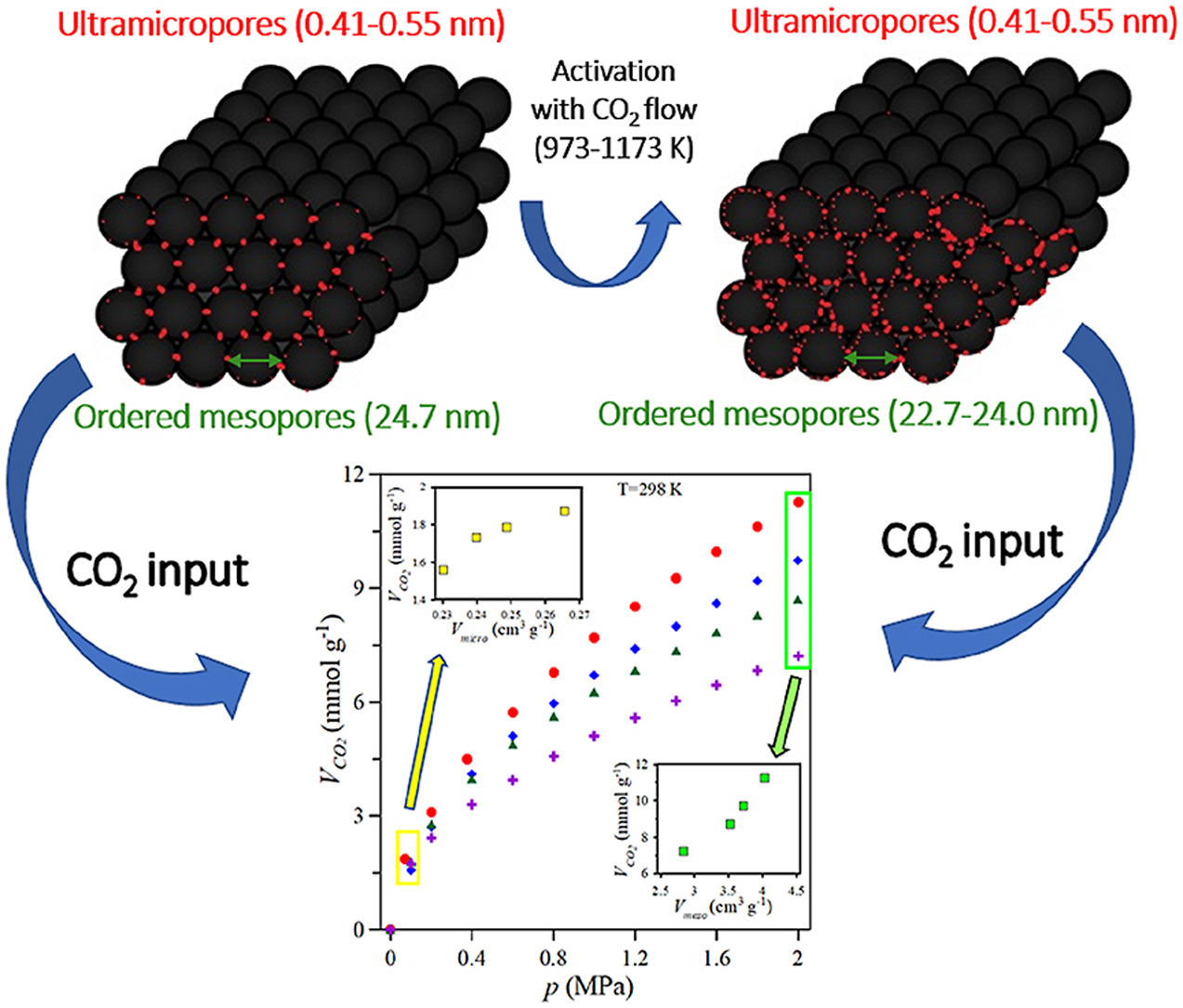
Three-dimensionally ordered micromesoporous carbon (3DOmm) has been studied recently as a potential sorbent for CO2 capture at higher pressures. However, the as-synthesized material usually has a low microporous fraction, which could significantly limit CO2 uptake. Herein, we showed that microporosity in 3DOmm carbon can be increased by physical activation, substantially improving its CO2 capture performance in the wide pressure range. The highly porous activated 3DOmm carbons, having amorphous structure, were prepared with ordered spherical mesopores of diameter 22.7–24.7 nm and ultramicropores in the walls of diameter 0.41–0.55 nm. Physical activation was performed by exposing the post-synthesized 3DOmm carbon to high temperatures of 973–1173 K under a CO2 flow for 30 min. The activation procedure did not show any visible destruction of the ordered mesoporous structure in the 3DOmm carbon. However, it had a significant impact on micropore and mesopore volumes and the specific surface area, which in turn affected the CO2 adsorption capacity of the carbons. Accordingly, after activation at 973 K, the micropore volume of 0.23 cm3/g, the mesopore volume of 3.70 cm3/g and the specific surface area of 1058 m2/g in the 3DOmm carbon increased to 0.27 cm3/g, 3.94 cm3/g and 1462 m2/g, respectively. Further increase in activation temperature led to decreasing pore values due to widening and shrinking of the thin carbon walls in the 3DOmm carbon. The 3DOmm carbon activated at 973 K had the highest micropore and mesopore volumes and the highest CO2 adsorption performance over the whole pressure range (3.19 mmol/g at 273 K and 100 kPa; 11.18 mmol/g at 298 K and 2 MPa and 0.38 mmol/g under flue 15% CO2/85% N2 gas conditions at 298 K). All 3DOmm carbons showed fast kinetics, high selectivity for CO2 over N2 (33.2–51.5), the excellent regenerative ability and isosteric heats (18.9–29.2 kJ/mol), indicating physical adsorption.
For more details see: https://doi.org/10.1016/j.mtsust.2023.100509
© 2021 Matematicko-fyzikální fakulta Univerzity Karlovy.
Všechna práva vyhrazena. | Cookies